Sensory systems must reduce the transmission of redundant information to function efficiently. One strategy is to continuously adjust the sensitivity of neurons to suppress responses to common features of the input while enhancing responses to new ones.
In this project we used 2-photon imaging of the synaptic transmitter glutamate. Taking advantage of the genetically encoded fluoresecent reporter of glutamate iGluSnFR we were able to image the synaptic inputs impinging upon retinal ganglion cells as well as the output of the same ganglion cells at their axon terminals.

We show that the synaptic terminals of retinal bipolar cells compute the orientation of edges and that the retina signals the orientation of edges by two distinct means. One population of retinal ganglion cells are statically tuned, receiving inputs from retinal bipolar cells that are all tuned to a single orientation. A second population of retinal ganglion cells receive a mix of inputs from retinal bipolar cells tuned to different orientations. These ganglion cells are able to respond transiently to a change in orientation yet mainatin sensitivity to any further change in orientation, a computation termed dynamic predictive coding.
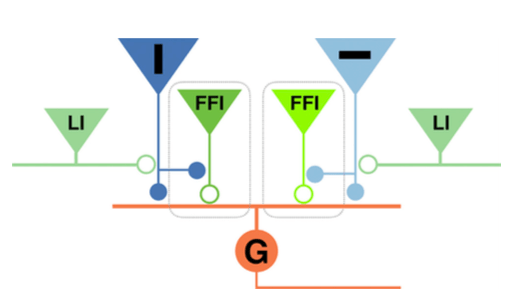
We also revealed key roles for inhibitory neurons within this circuit. Lateral inhibition is required for the computation of orientation by retinal bipolar cell synapses (see diagram). Whereas feedforward inhibition is required to generate a high-pass filter that enables ganglion cells carrying a dynamic predictive code to only transmits the initial activation of their different retinal bipolar cell inputs, thus removing redundancy. These results demonstrate how a dynamic predictive code can be implemented by circuit motifs common to many parts of the brain. The full paper is available here.
A Retinal Circuit Generating a Dynamic Predictive Code for Oriented Features
